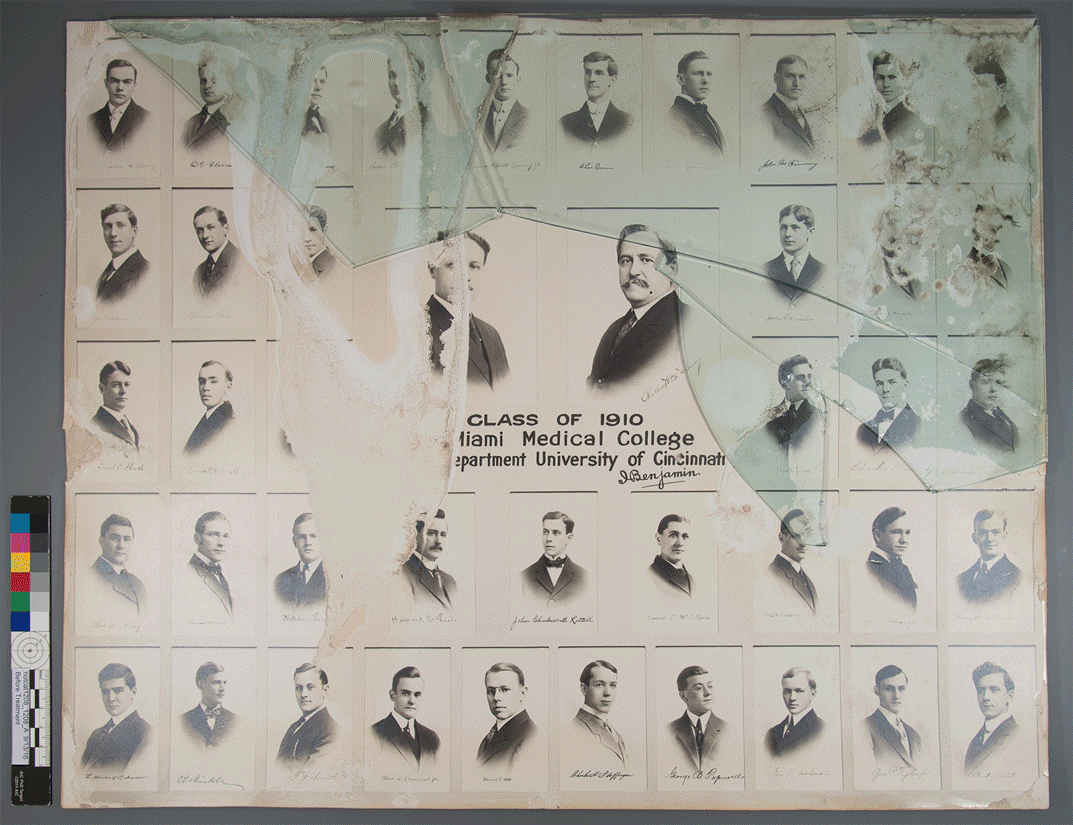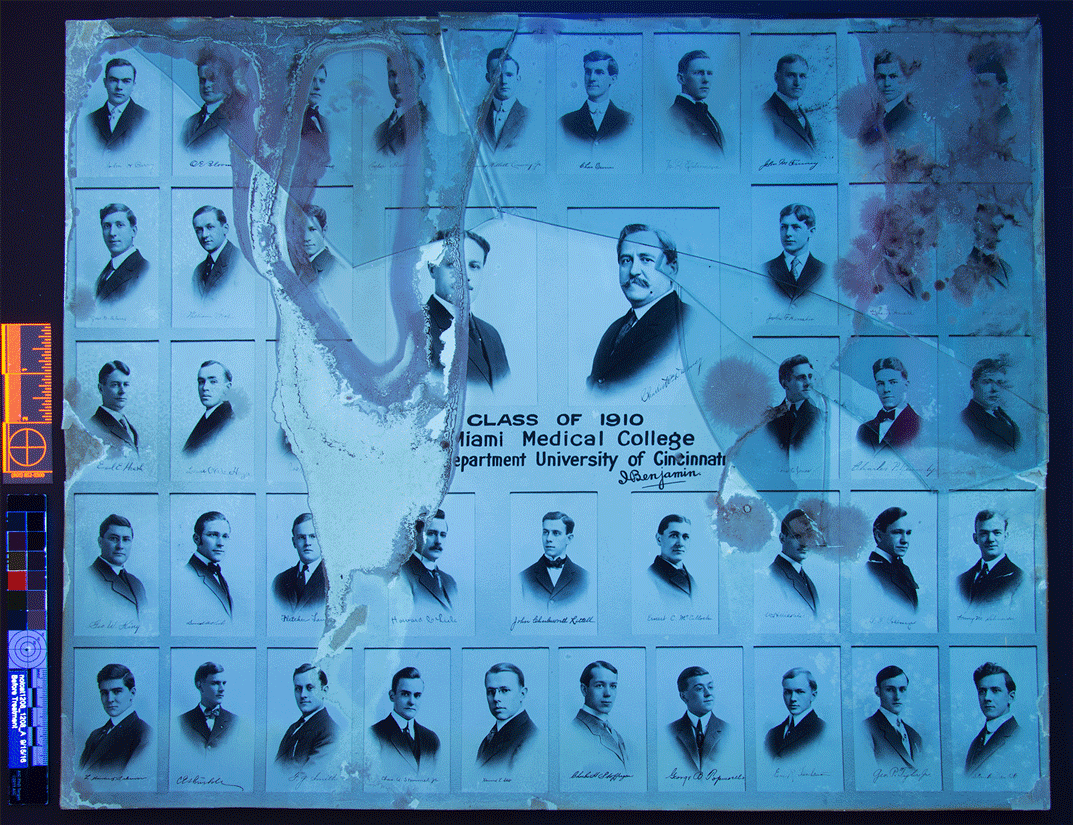Introduction and a Little Something About Halides
About two years ago, I set upon a mission to gain expertise in the area of identification and treatment of photographic materials. Under the guidance of our conservator, Ashleigh, I developed an education plan that was split between the theory of learning the ins and outs of photograph identification, and the hands-on work of treating pieces that came into the Lab. Of course, these two things go hand in hand. If you can’t identify a piece, you can’t treat it correctly, right?
Fast forward to last year. With the start of the pandemic and the transition to working from home, my education plan changed radically. If I’m not in the Lab, I can’t spend much time on treatment, so I had to get a little creative and work on other ways to learn more.
Enter the American Institute for Conservation’s self-study series on Photographic Chemistry for Preservation. It involves eight fairly in-depth units on silver-based analog photographs, how they are created, and as a consequence, how they age and deteriorate.
I am about halfway through the series; a triumph for me, as I have never been one for the study of chemistry. I will say that while it is still very technical, I’ve had a lot of good pegs to hang the information on, owing both to my earlier studies in photograph conservation and my personal history with film photography. It’s been a tremendous thing, viewing things that I learned as a photography student from a different angle. So far, it’s been a great journey.
In this series, I will share with you some of the most fascinating things that I’ve learned so far. My aim will be to keep the technical as simple as possible, for those of you who are like me, still coming to terms with the deeper science. The small bites help it all make sense, I promise. Hopefully, you’ll find it all as interesting as I have.
Before we can understand anything else, we need to talk about halides. What are those and why are they used in photography? Good questions! Halide salts are derived from halogens, which occupy group 7A (column 17) of the Periodic Table of Elements (see below.) Halide salts are used in photographic emulsions that are spread over a substrate (such as paper or film) before the substrate is exposed to light. The silver halides react to the light to form an image when developed.
I should note here that silver gelatin prints, albumen, and collodion photographs all utilize silver halides in their chemical composition. However, silver gelatin is unique among the three in that it is the only one that uses a true emulsion; in albumen and collodion coatings, the halides rest on the surface.

Photograph – silver gelatin process 
Photograph – albumin process 
Photograph – collodion process
In forming the silver gelatin emulsion, halide salts are combined with silver nitrate and water to form silver halides, the compound at the core of silver gelatin photography. Silver nitrate is pretty much universally used regardless of halide salt, as it is water soluble (it dissolves) but not too much so. The freed silver will look for a bond partner, and the halides in halide salt fits the bill. As a result, silver nitrate, when combined with a halide salt in water, will result in silver halide and a left over salt.
This reaction, which seems like a lot, I know, is referred to for our purposes as “The Emulsification Equation.” To refresh our memories a bit, an emulsification is a liquid (here, gelatin) that contains fine particles of another liquid (the silver halide) without fully combining. Think mayonnaise, or butter. (This isn’t perfectly analogous, as silver halides are crystalline solids and not liquid fats, but the basic idea is the same.)
Chemically speaking, that reaction looks like this:

As a quick reminder, Ag = silver, N = Nitrogen, O = Oxygen, K = Potassium, and Cl = Chlorine.
Now, if you’ll look at the image of the halogen column of the table below, you’ll see a number of options for salts to combine with silver nitrate. Older emulsions involved bromine or iodine; more modern emulsions tend toward chlorine. Crystals formed from silver chloride salts are much more uniform in structure, which makes its use outcomes much more predictable.
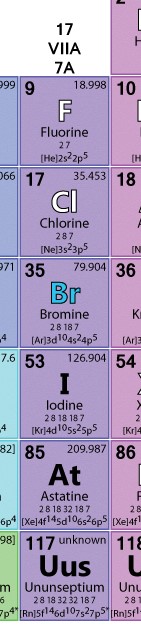
I’m sure you’ve noticed that we’ve got a couple of halogens unaccounted for, namely fluorine and astatine. Neither of these are used for this kind of work, and for good reason. Fluorine, for its part, is very water soluble. Very water soluble. To put it in perspective, sodium chloride (regular table salt) is about 35% water soluble. I’m sure that in the course of cooking, we’ve all dissolved salt in water, and you can recall how relatively simple that is to do, though not without some small effort. Well, fluorine salts are about 172% water soluble! You could use it for your emulsion, but moments after developing an image in a water-based solution, you’d see it dissolve before your eyes.
I’ll note here very briefly that chlorine, bromine, and iodine are also more soluble than table salt, but not nearly as much as fluorine, making them perfect partners for our silver ions.
Meanwhile, astatine is…well, it’s radioactive. I think you can see the problem with this one.
And there you have it, a short and hopefully painless explanation of the humble halide in silver-based photography. In the coming months, we’ll be looking at other fascinating aspects of halides and our Emulsification Equation.
Hyacinth Tucker (UCL) —- Bindery and Conservation Technician